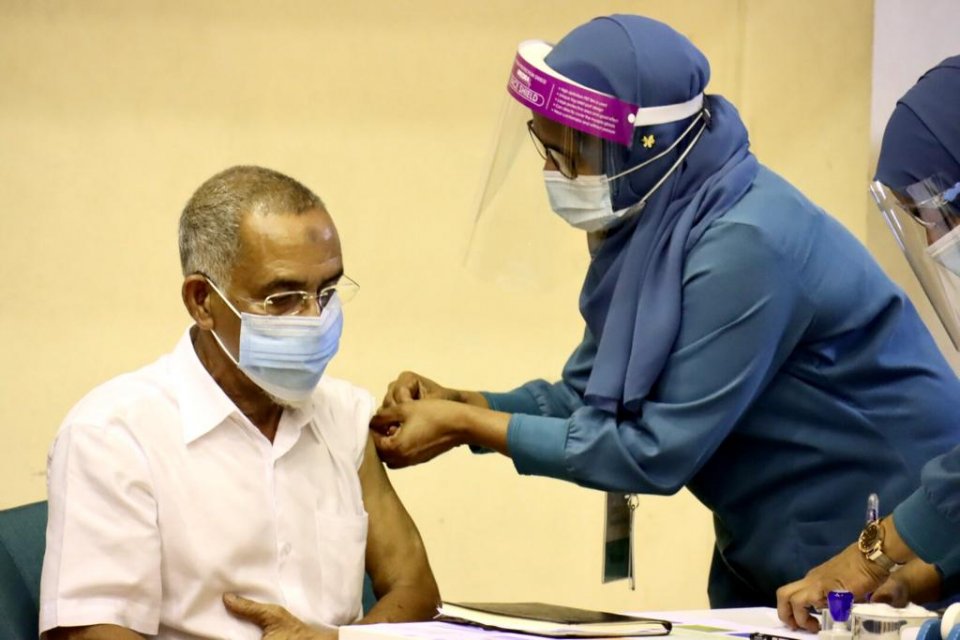
Health Protection Agency HPA has announced that the agency would be starting the second round of vaccination in the Greater Male' area tomorrow with Sinopharm vaccine donated by the Chinese government.
HPA said in a tweet that those who were not able to get their vaccine in the first round can now get vaccinated and that vaccination is open for everyone over 18 years of age.
The Agency shared vital information about the Chinese developed vaccine and said that Sinopharm is an inactivated vaccine. HPA said that it was developed by Sinopharm and Beijing Bio-Institute of Biological Products Co-Ltd of China.
HPA clarified that the Sinopharm vaccine has been approved for use in more than 20 countries across the world.
The Agency noted that children below 18 years of age, patients with active COVID-19, people with diseases or on medication that make their immune system weak as well as pregnant women cannot receive the Sinopharm vaccine.
HPA highlighted the common side effects of the vaccine as;
- Tenderness, pain, warmth, redness, itching, swelling, or bruising at the injection site
- Fatigue
- Headache
- Fever or Shivering
- Vomiting and/ or diarrhea
- Joint pain
- Muscle ache
However, HPA did note that side effects are normal signs that the recipient's body is building protection against COVID-19.
The Agency also said that people who are unwell (with or without fever) can receive the vaccine 3 days after symptoms resolve. It said that patients who test positive for COVID-19 can receive the Sinopharm vaccine 28 days from onset of symptoms, after recovery.
HPA added that people who have had an organ transplant may receive the vaccine 3 months after transplantation.
HPA highlighted that the Sinopharm is a two-dose vaccine and that the 2nd dose of the vaccine will be given to 3 to 4 weeks after the first dose. HPA added that anyone above the age of 18 can receive the Sinopharm vaccine.
The Agency had been administering the covishield vaccine donated by India earlier this year and has now administered the first dose of the vaccine to 218,912 individuals.